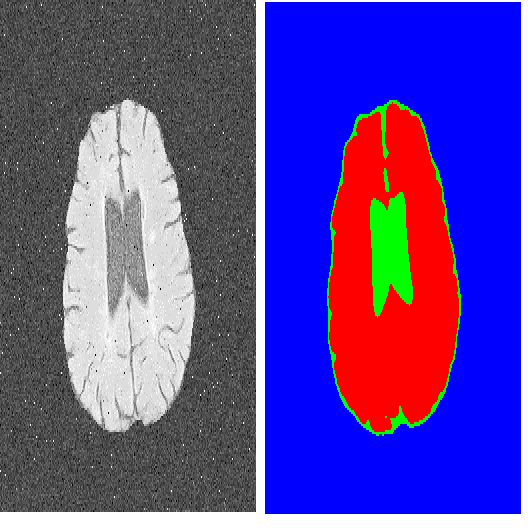
Learning to segmentation without large-scale samples is an inherent capability of human. Recently, Segment Anything Model (SAM) performs the significant zero-shot image segmentation, attracting considerable attention from the computer vision community. Here, we investigate the capability of SAM for medical image analysis, especially for multi-phase liver tumor segmentation (MPLiTS), in terms of prompts, data resolution, phases. Experimental results demonstrate that there might be a large gap between SAM and expected performance. Fortunately, the qualitative results show that SAM is a powerful annotation tool for the community of interactive medical image segmentation.
相關內容
The Segment Anything Model (SAM) has recently emerged as a groundbreaking model in the field of image segmentation. Nevertheless, both the original SAM and its medical adaptations necessitate slice-by-slice annotations, which directly increase the annotation workload with the size of the dataset. We propose MedLSAM to address this issue, ensuring a constant annotation workload irrespective of dataset size and thereby simplifying the annotation process. Our model introduces a few-shot localization framework capable of localizing any target anatomical part within the body. To achieve this, we develop a Localize Anything Model for 3D Medical Images (MedLAM), utilizing two self-supervision tasks: relative distance regression (RDR) and multi-scale similarity (MSS) across a comprehensive dataset of 14,012 CT scans. We then establish a methodology for accurate segmentation by integrating MedLAM with SAM. By annotating only six extreme points across three directions on a few templates, our model can autonomously identify the target anatomical region on all data scheduled for annotation. This allows our framework to generate a 2D bounding box for every slice of the image, which are then leveraged by SAM to carry out segmentations. We conducted experiments on two 3D datasets covering 38 organs and found that MedLSAM matches the performance of SAM and its medical adaptations while requiring only minimal extreme point annotations for the entire dataset. Furthermore, MedLAM has the potential to be seamlessly integrated with future 3D SAM models, paving the way for enhanced performance. Our code is public at \href{//github.com/openmedlab/MedLSAM}{//github.com/openmedlab/MedLSAM}.
Semi-supervised learning has demonstrated great potential in medical image segmentation by utilizing knowledge from unlabeled data. However, most existing approaches do not explicitly capture high-level semantic relations between distant regions, which limits their performance. In this paper, we focus on representation learning for semi-supervised learning, by developing a novel Multi-Scale Cross Supervised Contrastive Learning (MCSC) framework, to segment structures in medical images. We jointly train CNN and Transformer models, regularising their features to be semantically consistent across different scales. Our approach contrasts multi-scale features based on ground-truth and cross-predicted labels, in order to extract robust feature representations that reflect intra- and inter-slice relationships across the whole dataset. To tackle class imbalance, we take into account the prevalence of each class to guide contrastive learning and ensure that features adequately capture infrequent classes. Extensive experiments on two multi-structure medical segmentation datasets demonstrate the effectiveness of MCSC. It not only outperforms state-of-the-art semi-supervised methods by more than 3.0% in Dice, but also greatly reduces the performance gap with fully supervised methods.
Despite that the segment anything model (SAM) achieved impressive results on general-purpose semantic segmentation with strong generalization ability on daily images, its demonstrated performance on medical image segmentation is less precise and not stable, especially when dealing with tumor segmentation tasks that involve objects of small sizes, irregular shapes, and low contrast. Notably, the original SAM architecture is designed for 2D natural images, therefore would not be able to extract the 3D spatial information from volumetric medical data effectively. In this paper, we propose a novel adaptation method for transferring SAM from 2D to 3D for promptable medical image segmentation. Through a holistically designed scheme for architecture modification, we transfer the SAM to support volumetric inputs while retaining the majority of its pre-trained parameters for reuse. The fine-tuning process is conducted in a parameter-efficient manner, wherein most of the pre-trained parameters remain frozen, and only a few lightweight spatial adapters are introduced and tuned. Regardless of the domain gap between natural and medical data and the disparity in the spatial arrangement between 2D and 3D, the transformer trained on natural images can effectively capture the spatial patterns present in volumetric medical images with only lightweight adaptations. We conduct experiments on four open-source tumor segmentation datasets, and with a single click prompt, our model can outperform domain state-of-the-art medical image segmentation models on 3 out of 4 tasks, specifically by 8.25%, 29.87%, and 10.11% for kidney tumor, pancreas tumor, colon cancer segmentation, and achieve similar performance for liver tumor segmentation. We also compare our adaptation method with existing popular adapters, and observed significant performance improvement on most datasets.
Image restoration is a low-level visual task, and most CNN methods are designed as black boxes, lacking transparency and intrinsic aesthetics. Many unsupervised approaches ignore the degradation of visible information in low-light scenes, which will seriously affect the aggregation of complementary information and also make the fusion algorithm unable to produce satisfactory fusion results under extreme conditions. In this paper, we propose Enlighten-anything, which is able to enhance and fuse the semantic intent of SAM segmentation with low-light images to obtain fused images with good visual perception. The generalization ability of unsupervised learning is greatly improved, and experiments on LOL dataset are conducted to show that our method improves 3db in PSNR over baseline and 8 in SSIM. Zero-shot learning of SAM introduces a powerful aid for unsupervised low-light enhancement. The source code of Enlighten Anything can be obtained from //github.com/zhangbaijin/enlighten-anything
In this study, the main objective is to develop an algorithm capable of identifying and delineating tumor regions in breast ultrasound (BUS) and mammographic images. The technique employs two advanced deep learning architectures, namely U-Net and pretrained SAM, for tumor segmentation. The U-Net model is specifically designed for medical image segmentation and leverages its deep convolutional neural network framework to extract meaningful features from input images. On the other hand, the pretrained SAM architecture incorporates a mechanism to capture spatial dependencies and generate segmentation results. Evaluation is conducted on a diverse dataset containing annotated tumor regions in BUS and mammographic images, covering both benign and malignant tumors. This dataset enables a comprehensive assessment of the algorithm's performance across different tumor types. Results demonstrate that the U-Net model outperforms the pretrained SAM architecture in accurately identifying and segmenting tumor regions in both BUS and mammographic images. The U-Net exhibits superior performance in challenging cases involving irregular shapes, indistinct boundaries, and high tumor heterogeneity. In contrast, the pretrained SAM architecture exhibits limitations in accurately identifying tumor areas, particularly for malignant tumors and objects with weak boundaries or complex shapes. These findings highlight the importance of selecting appropriate deep learning architectures tailored for medical image segmentation. The U-Net model showcases its potential as a robust and accurate tool for tumor detection, while the pretrained SAM architecture suggests the need for further improvements to enhance segmentation performance.
Most deep learning-based models for speech enhancement have mainly focused on estimating the magnitude of spectrogram while reusing the phase from noisy speech for reconstruction. This is due to the difficulty of estimating the phase of clean speech. To improve speech enhancement performance, we tackle the phase estimation problem in three ways. First, we propose Deep Complex U-Net, an advanced U-Net structured model incorporating well-defined complex-valued building blocks to deal with complex-valued spectrograms. Second, we propose a polar coordinate-wise complex-valued masking method to reflect the distribution of complex ideal ratio masks. Third, we define a novel loss function, weighted source-to-distortion ratio (wSDR) loss, which is designed to directly correlate with a quantitative evaluation measure. Our model was evaluated on a mixture of the Voice Bank corpus and DEMAND database, which has been widely used by many deep learning models for speech enhancement. Ablation experiments were conducted on the mixed dataset showing that all three proposed approaches are empirically valid. Experimental results show that the proposed method achieves state-of-the-art performance in all metrics, outperforming previous approaches by a large margin.
The U-Net was presented in 2015. With its straight-forward and successful architecture it quickly evolved to a commonly used benchmark in medical image segmentation. The adaptation of the U-Net to novel problems, however, comprises several degrees of freedom regarding the exact architecture, preprocessing, training and inference. These choices are not independent of each other and substantially impact the overall performance. The present paper introduces the nnU-Net ('no-new-Net'), which refers to a robust and self-adapting framework on the basis of 2D and 3D vanilla U-Nets. We argue the strong case for taking away superfluous bells and whistles of many proposed network designs and instead focus on the remaining aspects that make out the performance and generalizability of a method. We evaluate the nnU-Net in the context of the Medical Segmentation Decathlon challenge, which measures segmentation performance in ten disciplines comprising distinct entities, image modalities, image geometries and dataset sizes, with no manual adjustments between datasets allowed. At the time of manuscript submission, nnU-Net achieves the highest mean dice scores across all classes and seven phase 1 tasks (except class 1 in BrainTumour) in the online leaderboard of the challenge.

We propose a novel attention gate (AG) model for medical imaging that automatically learns to focus on target structures of varying shapes and sizes. Models trained with AGs implicitly learn to suppress irrelevant regions in an input image while highlighting salient features useful for a specific task. This enables us to eliminate the necessity of using explicit external tissue/organ localisation modules of cascaded convolutional neural networks (CNNs). AGs can be easily integrated into standard CNN architectures such as the U-Net model with minimal computational overhead while increasing the model sensitivity and prediction accuracy. The proposed Attention U-Net architecture is evaluated on two large CT abdominal datasets for multi-class image segmentation. Experimental results show that AGs consistently improve the prediction performance of U-Net across different datasets and training sizes while preserving computational efficiency. The code for the proposed architecture is publicly available.

Recent advances in 3D fully convolutional networks (FCN) have made it feasible to produce dense voxel-wise predictions of volumetric images. In this work, we show that a multi-class 3D FCN trained on manually labeled CT scans of several anatomical structures (ranging from the large organs to thin vessels) can achieve competitive segmentation results, while avoiding the need for handcrafting features or training class-specific models. To this end, we propose a two-stage, coarse-to-fine approach that will first use a 3D FCN to roughly define a candidate region, which will then be used as input to a second 3D FCN. This reduces the number of voxels the second FCN has to classify to ~10% and allows it to focus on more detailed segmentation of the organs and vessels. We utilize training and validation sets consisting of 331 clinical CT images and test our models on a completely unseen data collection acquired at a different hospital that includes 150 CT scans, targeting three anatomical organs (liver, spleen, and pancreas). In challenging organs such as the pancreas, our cascaded approach improves the mean Dice score from 68.5 to 82.2%, achieving the highest reported average score on this dataset. We compare with a 2D FCN method on a separate dataset of 240 CT scans with 18 classes and achieve a significantly higher performance in small organs and vessels. Furthermore, we explore fine-tuning our models to different datasets. Our experiments illustrate the promise and robustness of current 3D FCN based semantic segmentation of medical images, achieving state-of-the-art results. Our code and trained models are available for download: //github.com/holgerroth/3Dunet_abdomen_cascade.
Image segmentation is considered to be one of the critical tasks in hyperspectral remote sensing image processing. Recently, convolutional neural network (CNN) has established itself as a powerful model in segmentation and classification by demonstrating excellent performances. The use of a graphical model such as a conditional random field (CRF) contributes further in capturing contextual information and thus improving the segmentation performance. In this paper, we propose a method to segment hyperspectral images by considering both spectral and spatial information via a combined framework consisting of CNN and CRF. We use multiple spectral cubes to learn deep features using CNN, and then formulate deep CRF with CNN-based unary and pairwise potential functions to effectively extract the semantic correlations between patches consisting of three-dimensional data cubes. Effective piecewise training is applied in order to avoid the computationally expensive iterative CRF inference. Furthermore, we introduce a deep deconvolution network that improves the segmentation masks. We also introduce a new dataset and experimented our proposed method on it along with several widely adopted benchmark datasets to evaluate the effectiveness of our method. By comparing our results with those from several state-of-the-art models, we show the promising potential of our method.