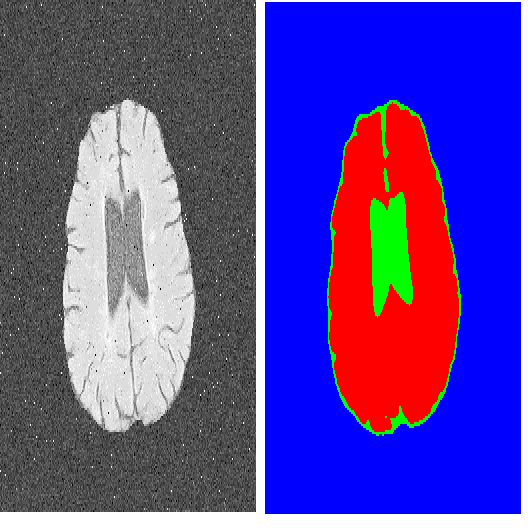
Many eye diseases like Diabetic Macular Edema (DME), Age-related Macular Degeneration (AMD), and Glaucoma manifest in the retina, can cause irreversible blindness or severely impair the central version. The Optical Coherence Tomography (OCT), a 3D scan of the retina with high qualitative information about the retinal morphology, can be used to diagnose and monitor changes in the retinal anatomy. Many Deep Learning (DL) methods have shared the success of developing an automated tool to monitor pathological changes in the retina. However, the success of these methods depend mainly on large datasets. To address the challenge from very small and limited datasets, we proposed a DL architecture termed CoNet (Coherent Network) for joint segmentation of layers and fluids in retinal OCT images on very small datasets (less than a hundred training samples). The proposed model was evaluated on the publicly available Duke DME dataset consisting of 110 B-Scans from 10 patients suffering from DME. Experimental results show that the proposed model outperformed both the human experts' annotation and the current state-of-the-art architectures by a clear margin with a mean Dice Score of 88% when trained on 55 images without any data augmentation.
相關內容
Reducing the quantity of annotations required for supervised training is vital when labels are scarce and costly. This reduction is particularly important for semantic segmentation tasks involving 3D datasets, which are often significantly smaller and more challenging to annotate than their image-based counterparts. Self-supervised pre-training on unlabelled data is one way to reduce the amount of manual annotations needed. Previous work has focused on pre-training with point clouds exclusively. While useful, this approach often requires two or more registered views. In the present work, we combine image and point cloud modalities by first learning self-supervised image features and then using these features to train a 3D model. By incorporating image data, which is often included in many 3D datasets, our pre-training method only requires a single scan of a scene and can be applied to cases where localization information is unavailable. We demonstrate that our pre-training approach, despite using single scans, achieves comparable performance to other multi-scan, point cloud-only methods.
Although the preservation of shape continuity and physiological anatomy is a natural assumption in the segmentation of medical images, it is often neglected by deep learning methods that mostly aim for the statistical modeling of input data as pixels rather than interconnected structures. In biological structures, however, organs are not separate entities; for example, in reality, a severed vessel is an indication of an underlying problem, but traditional segmentation models are not designed to strictly enforce the continuity of anatomy, potentially leading to inaccurate medical diagnoses. To address this issue, we propose a graph-based approach that enforces the continuity and connectivity of anatomical topology in medical images. Our method encodes the continuity of shapes as a graph constraint, ensuring that the network's predictions maintain this continuity. We evaluate our method on two public benchmarks on retinal vessel segmentation, showing significant improvements in connectivity metrics compared to traditional methods while getting better or on-par performance on segmentation metrics.
Although purely transformer-based architectures showed promising performance in many computer vision tasks, many hybrid models consisting of CNN and transformer blocks are introduced to fit more specialized tasks. Nevertheless, despite the performance gain of both pure and hybrid transformer-based architectures compared to CNNs in medical imaging segmentation, their high training cost and complexity make it challenging to use them in real scenarios. In this work, we propose simple architectures based on purely convolutional layers, and show that by just taking advantage of the attention map visualizations obtained from a self-supervised pretrained vision transformer network (e.g., DINO) one can outperform complex transformer-based networks with much less computation costs. The proposed architecture is composed of two encoder branches with the original image as input in one branch and the attention map visualizations of the same image from multiple self-attention heads from a pre-trained DINO model (as multiple channels) in the other branch. The results of our experiments on two publicly available medical imaging datasets show that the proposed pipeline outperforms U-Net and the state-of-the-art medical image segmentation models.
Deep learning models have demonstrated remarkable success in multi-organ segmentation but typically require large-scale datasets with all organs of interest annotated. However, medical image datasets are often low in sample size and only partially labeled, i.e., only a subset of organs are annotated. Therefore, it is crucial to investigate how to learn a unified model on the available partially labeled datasets to leverage their synergistic potential. In this paper, we empirically and systematically study the partial-label segmentation with in-depth analyses on the existing approaches and identify three distinct types of supervision signals, including two signals derived from ground truth and one from pseudo label. We propose a novel training framework termed COSST, which effectively and efficiently integrates comprehensive supervision signals with self-training. Concretely, we first train an initial unified model using two ground truth-based signals and then iteratively incorporate the pseudo label signal to the initial model using self-training. To mitigate performance degradation caused by unreliable pseudo labels, we assess the reliability of pseudo labels via outlier detection in latent space and exclude the most unreliable pseudo labels from each self-training iteration. Extensive experiments are conducted on six CT datasets for three partial-label segmentation tasks. Experimental results show that our proposed COSST achieves significant improvement over the baseline method, i.e., individual networks trained on each partially labeled dataset. Compared to the state-of-the-art partial-label segmentation methods, COSST demonstrates consistent superior performance on various segmentation tasks and with different training data size.
Medical image segmentation is a fundamental and critical step in many image-guided clinical approaches. Recent success of deep learning-based segmentation methods usually relies on a large amount of labeled data, which is particularly difficult and costly to obtain especially in the medical imaging domain where only experts can provide reliable and accurate annotations. Semi-supervised learning has emerged as an appealing strategy and been widely applied to medical image segmentation tasks to train deep models with limited annotations. In this paper, we present a comprehensive review of recently proposed semi-supervised learning methods for medical image segmentation and summarized both the technical novelties and empirical results. Furthermore, we analyze and discuss the limitations and several unsolved problems of existing approaches. We hope this review could inspire the research community to explore solutions for this challenge and further promote the developments in medical image segmentation field.
We study the problem of efficient semantic segmentation for large-scale 3D point clouds. By relying on expensive sampling techniques or computationally heavy pre/post-processing steps, most existing approaches are only able to be trained and operate over small-scale point clouds. In this paper, we introduce RandLA-Net, an efficient and lightweight neural architecture to directly infer per-point semantics for large-scale point clouds. The key to our approach is to use random point sampling instead of more complex point selection approaches. Although remarkably computation and memory efficient, random sampling can discard key features by chance. To overcome this, we introduce a novel local feature aggregation module to progressively increase the receptive field for each 3D point, thereby effectively preserving geometric details. Extensive experiments show that our RandLA-Net can process 1 million points in a single pass with up to 200X faster than existing approaches. Moreover, our RandLA-Net clearly surpasses state-of-the-art approaches for semantic segmentation on two large-scale benchmarks Semantic3D and SemanticKITTI.
The U-Net was presented in 2015. With its straight-forward and successful architecture it quickly evolved to a commonly used benchmark in medical image segmentation. The adaptation of the U-Net to novel problems, however, comprises several degrees of freedom regarding the exact architecture, preprocessing, training and inference. These choices are not independent of each other and substantially impact the overall performance. The present paper introduces the nnU-Net ('no-new-Net'), which refers to a robust and self-adapting framework on the basis of 2D and 3D vanilla U-Nets. We argue the strong case for taking away superfluous bells and whistles of many proposed network designs and instead focus on the remaining aspects that make out the performance and generalizability of a method. We evaluate the nnU-Net in the context of the Medical Segmentation Decathlon challenge, which measures segmentation performance in ten disciplines comprising distinct entities, image modalities, image geometries and dataset sizes, with no manual adjustments between datasets allowed. At the time of manuscript submission, nnU-Net achieves the highest mean dice scores across all classes and seven phase 1 tasks (except class 1 in BrainTumour) in the online leaderboard of the challenge.
Deep neural network architectures have traditionally been designed and explored with human expertise in a long-lasting trial-and-error process. This process requires huge amount of time, expertise, and resources. To address this tedious problem, we propose a novel algorithm to optimally find hyperparameters of a deep network architecture automatically. We specifically focus on designing neural architectures for medical image segmentation task. Our proposed method is based on a policy gradient reinforcement learning for which the reward function is assigned a segmentation evaluation utility (i.e., dice index). We show the efficacy of the proposed method with its low computational cost in comparison with the state-of-the-art medical image segmentation networks. We also present a new architecture design, a densely connected encoder-decoder CNN, as a strong baseline architecture to apply the proposed hyperparameter search algorithm. We apply the proposed algorithm to each layer of the baseline architectures. As an application, we train the proposed system on cine cardiac MR images from Automated Cardiac Diagnosis Challenge (ACDC) MICCAI 2017. Starting from a baseline segmentation architecture, the resulting network architecture obtains the state-of-the-art results in accuracy without performing any trial-and-error based architecture design approaches or close supervision of the hyperparameters changes.
In this paper, we focus on three problems in deep learning based medical image segmentation. Firstly, U-net, as a popular model for medical image segmentation, is difficult to train when convolutional layers increase even though a deeper network usually has a better generalization ability because of more learnable parameters. Secondly, the exponential ReLU (ELU), as an alternative of ReLU, is not much different from ReLU when the network of interest gets deep. Thirdly, the Dice loss, as one of the pervasive loss functions for medical image segmentation, is not effective when the prediction is close to ground truth and will cause oscillation during training. To address the aforementioned three problems, we propose and validate a deeper network that can fit medical image datasets that are usually small in the sample size. Meanwhile, we propose a new loss function to accelerate the learning process and a combination of different activation functions to improve the network performance. Our experimental results suggest that our network is comparable or superior to state-of-the-art methods.

Recent advances in 3D fully convolutional networks (FCN) have made it feasible to produce dense voxel-wise predictions of volumetric images. In this work, we show that a multi-class 3D FCN trained on manually labeled CT scans of several anatomical structures (ranging from the large organs to thin vessels) can achieve competitive segmentation results, while avoiding the need for handcrafting features or training class-specific models. To this end, we propose a two-stage, coarse-to-fine approach that will first use a 3D FCN to roughly define a candidate region, which will then be used as input to a second 3D FCN. This reduces the number of voxels the second FCN has to classify to ~10% and allows it to focus on more detailed segmentation of the organs and vessels. We utilize training and validation sets consisting of 331 clinical CT images and test our models on a completely unseen data collection acquired at a different hospital that includes 150 CT scans, targeting three anatomical organs (liver, spleen, and pancreas). In challenging organs such as the pancreas, our cascaded approach improves the mean Dice score from 68.5 to 82.2%, achieving the highest reported average score on this dataset. We compare with a 2D FCN method on a separate dataset of 240 CT scans with 18 classes and achieve a significantly higher performance in small organs and vessels. Furthermore, we explore fine-tuning our models to different datasets. Our experiments illustrate the promise and robustness of current 3D FCN based semantic segmentation of medical images, achieving state-of-the-art results. Our code and trained models are available for download: //github.com/holgerroth/3Dunet_abdomen_cascade.